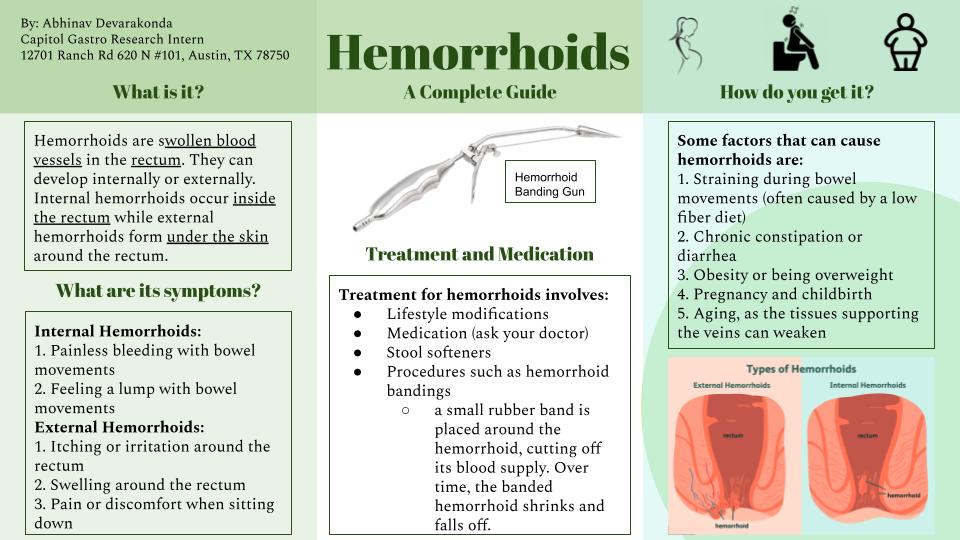
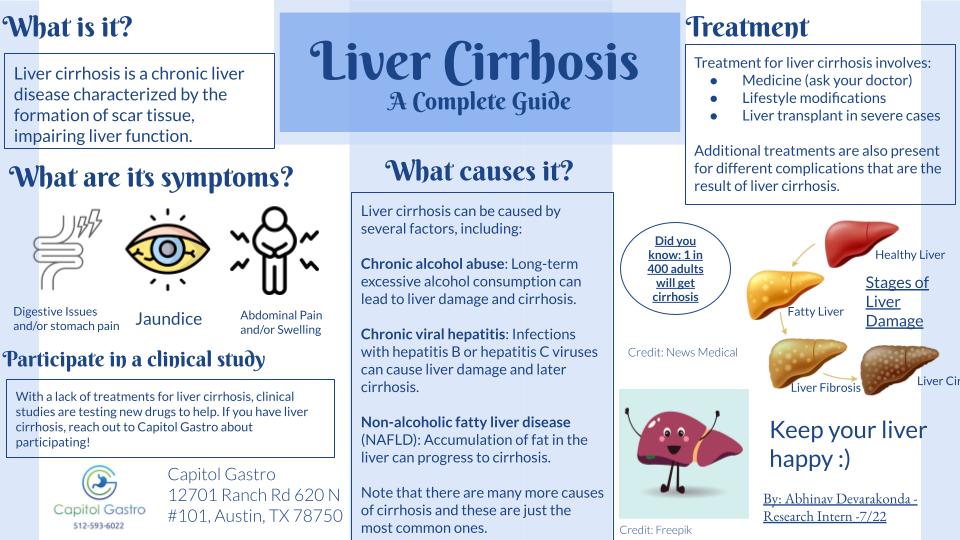
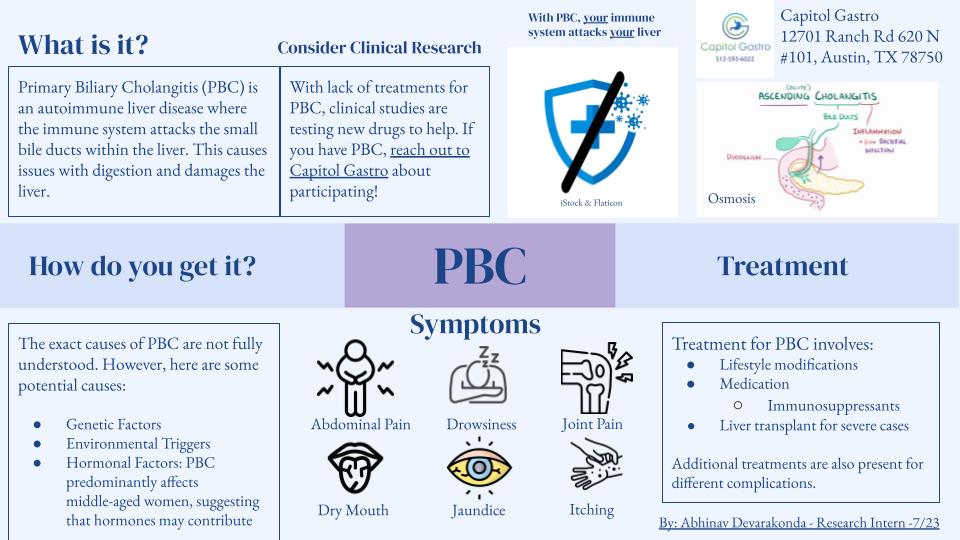
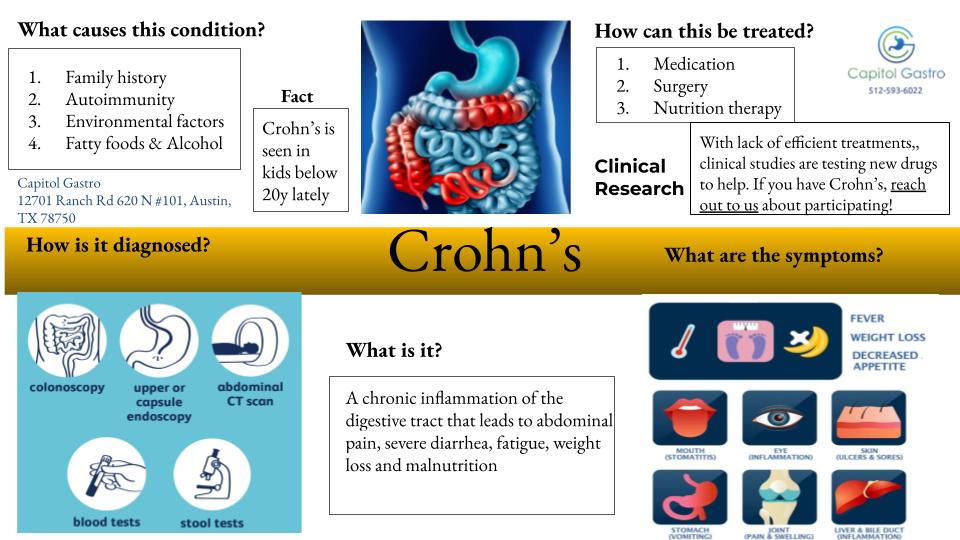
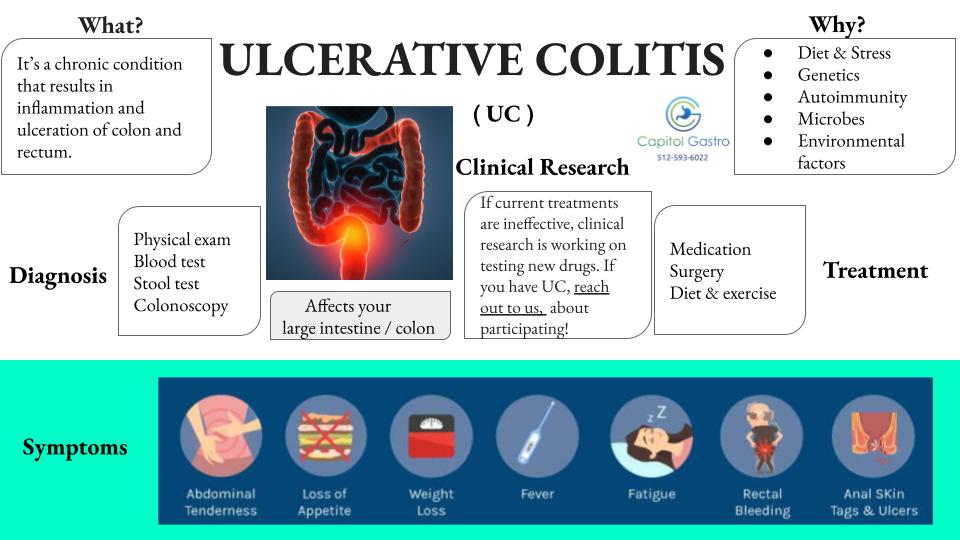
CLINICAL RESEARCH
Capitol Gastro’s Research Division, named as Accelemed Research Institute or AMEL MED LLC has contributed to clinical research since 2017 by participating in trials that helped to test medication for Crohn’s, Colorectal 7 Cervical Cancer, Liver Fibrosis, NASH (Non-Alcoholic Steatohepatitis), Ulcerative colitis. Our gastroenterologist, Dr. Masi Khaja has done investigation over 15 studies till now.
Accelemed research institute conducts studies as a reliable alternative to standard GI treatment methodologies for various medical diagnoses. Patients who do not show any improvement or have experienced side effects with the current medication are welcome to join the clinical trials. Interested participants are always given preference to become part of our research.
What to Expect from a Clinical Trial?
The clinical trial/study will be conducted locally in Austin, TX. The research participants will be provided with details of the study, duration, related procedures, visit schedule, and medication. All the above-mentioned procedures are compensated, will not be billed to the participants.
You will not be charged anything for participating in the clinical trial. All the study related expenses will be taken care by the sponsor.
Side effects of the drug, Allot time for Treatment visits (In-person or remote)
The ICH GCP guidelines are HIPAA compliant; include patient safety and well-being as the top priority in clinical trials.
ONGOING CLINICAL TRIALS
ONGOING CLINICAL TRIALS
Liver Cirrhosis Study – ENROLLING
Primary Biliary Cholangitis (PBC) with Itch Study – ENROLLING
Ulcerative Colitis study – WILL OPEN SOON
Crohn’s Disease Study1 – WILL OPEN SOON
Crohn’s Disease Study2 – ON HOLD
Severe Crohn’s Disease Study – CLOSED
CLINICAL-TRIALS-SYNOPSYS